“Clinical Trial Design”
Best Practice Hour Seminar Series
June 18, 2024
12:00 PM - 1:00 PM
Location
Virtual via Zoom
Calendar
Download iCal File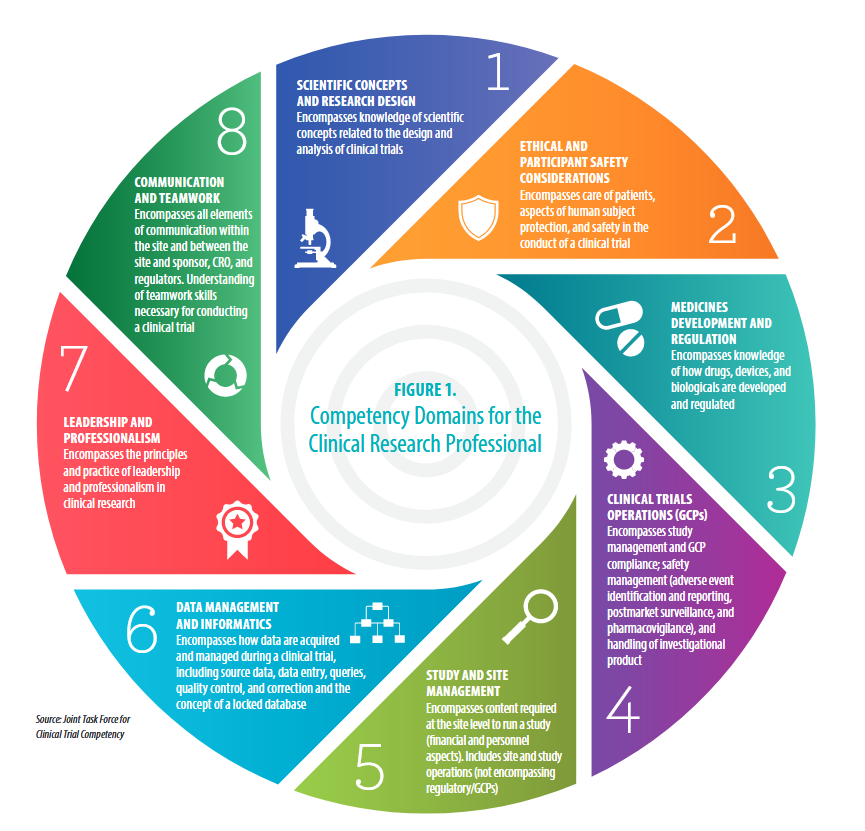
https://clic-ctsa.org/publications/ecrptq-competencies
Description:
This presentation will be an overview of clinical trial designs, with an emphasis on the design of FDA regulated clinical trials.
Speaker:
Darlene Kitterman, MBA
Director, Clinical Trials Office
University of Illinois Cancer Center
Competency Area:
#1: Scientific Concepts & Research Design
Register for Zoom login details
Date posted
Mar 4, 2024
Date updated
May 28, 2024